WHAT WAS CLAIMED
A vaccine safety body has recorded 20 times more COVID jab adverse reactions than the government’s Therapeutic Goods Administration.
OUR VERDICT
Misleading. The two reporting systems use different measures to gather different types of adverse reaction data. They are not comparable.
A Facebook video claims an Australian vaccine safety body has recorded significantly more adverse reactions to the COVID-19 jabs than the federal government's medical regulatory agency.
Mike Palmer, host of a legal advocacy livestream on the Know Your Rights Group Facebook page, said AusVaxSafety had reported 20 times more COVID vaccine side effects than the Therapeutic Goods Administration (TGA).
But his claim is misleading and missing important context. The two reporting systems gather different types of data using different methods.
One records mostly mild reactions such as nausea and fatigue, while the other reports more serious but less common side effects.
In the video (screenshot here) posted on December 8, 2022, Mr Palmer says the AusVaxSafety report from November 28 was "produced three days earlier than the TGA report, yet the numbers are more than 20 times higher" (video mark 27min 29sec).
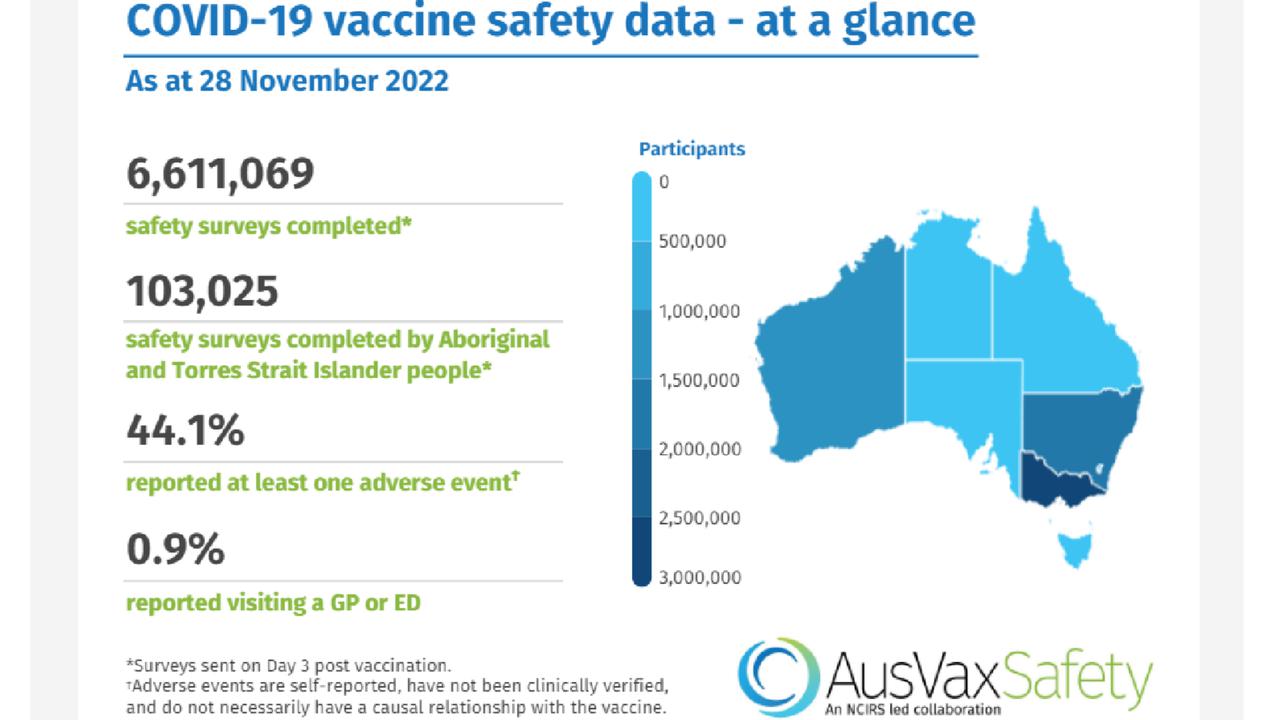
He tells his followers: "How does the TGA get away with reporting 137,000 adverse reactions when AusVaxSafety data reports 2.915 million adverse reactions?"
Implying underhand conduct, he adds: "Somewhere, somebody is not telling us the truth" (28min 22sec).
Mr Palmer arrives at the 2.915 million figure as 44.1 per cent of the 6,611,069 individuals surveyed by AusVaxSafety reported at least one adverse event.
His numbers are correct, but his claim is missing important context about the data collected by the two organisations.
An independent expert, as well as representatives from the TGA and AusVaxSafety, told AAP FactCheck that the two bodies intentionally record different kinds of data.
Associate Professor James Trauer, head of epidemiological modelling at Monash University, said the different datasets combined paint a fuller picture of vaccine side effects in Australia.
"The two surveillance systems provide different data on adverse [vaccine] events and together complement each other to provide a comprehensive surveillance system," Assoc Prof Trauer said in an email.

He explained the TGA is a passive system that collects unsolicited reports of adverse reactions. This means anyone can report a suspected side effect to the TGA after they've received a vaccine, without being prompted.
Individuals can report reactions to the TGA over the phone, via email or via an online form.
Health professionals and pharmaceutical companies can also send reports about adverse reactions in individuals.
He said because the TGA gathers spontaneous reports, its data usually contains information about rarer and more serious side effects such as myocarditis, pericarditis and blood clotting.
"These adverse events may also be reported to AusVaxSafety, but are more likely to be reported to the TGA as they are usually identified by health professionals," a TGA representative told AAP FactCheck in an email.
AusVaxSafety, on the other hand, is a surveillance system that actively seeks data on any vaccine side effect.
When someone gets a vaccine, they can receive a follow-up text or email from AusVaxSafety. More than 6.6 million surveys have been sent out at the time of writing.
"As the survey is sent directly to an individual and only takes minutes to complete, recipients may be more likely to report common expected adverse events (such as pain at injection site or headache) when compared to filling in a form to the TGA," an AusVaxSafety representative told AAP FactCheck in an email.
Assoc Prof Trauer agreed. He said: "By actively asking participants about their experience, there is a higher rate of reporting to AusVaxSafety – and importantly they are encouraged to report minor and short-lived adverse events such as soreness at the injection sites, headache and tiredness – which are also very common after vaccination."
As a result he said AusVaxSafety records significantly more mild adverse reactions, while the TGA generally reports fewer, but more serious, side effects.
He said the different systems were necessary and beneficial.
"Far from being concerned, Australians should be reassured by these results," he said.
"It's really a good thing that we have multiple systems for detecting and describing adverse events that occur after vaccination."
He added: "The systems are very different in the way they operate, so we would absolutely expect that the numbers we get back from each of them are different."
AAP FactCheck has previously debunked other claims from Mr Palmer and Know Your Rights (see here, here, here, here, here and here).
The Verdict
The claim that AusVaxSafety has recorded 20 times more adverse reactions to the COVID vaccine than the Therapeutic Goods Administration is misleading, as it is missing key context on how the data is collected and what data is collected.
Representatives from both organisations and an independent expert told AAP FactCheck that the two bodies record vaccine side effect data in different ways in order to comprehensively detect adverse reactions, ranging from mild and expected side effects to more serious but less common reactions.
Their respective figures are not and should not be comparable.
Misleading – The claim is accurate in parts but information has also been presented incorrectly, out of context or omitted.
AAP FactCheck is an accredited member of the International Fact-Checking Network. To keep up with our latest fact checks, follow us on Facebook, Twitter and Instagram.