The Statement
A Facebook post claiming to show a photo of a new vaccine for COVID-19 is spreading false hope as the economic, social and health impacts of the coronavirus deepen in Australia.
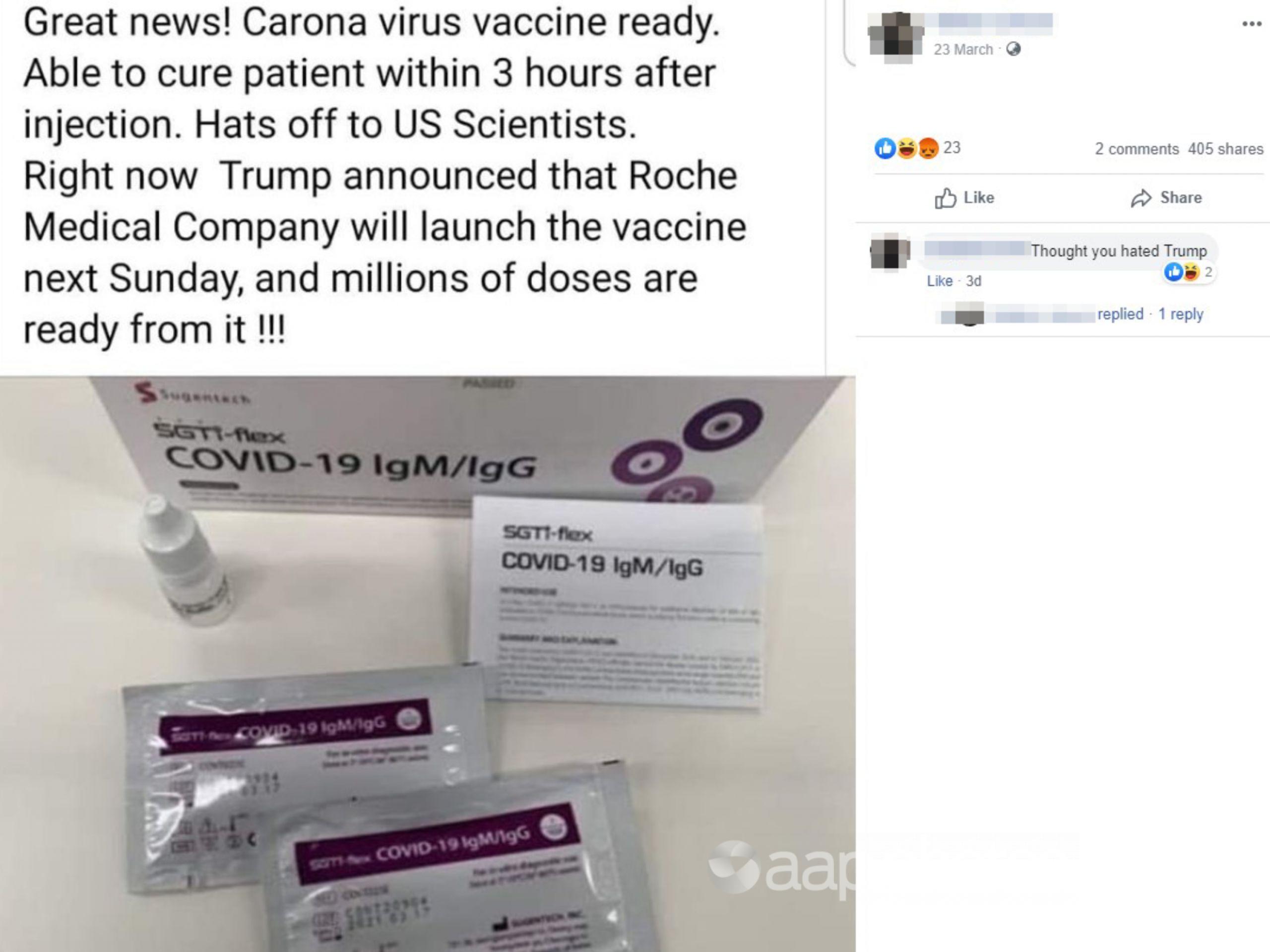
The March 23 post features a photograph of a pharmaceutical product, with a box, sachets and dropper bottle featuring the label “COVID-19 IgM-IgG”. The packaging is branded with the logo “Sugentech”.
The caption accompanying the image reads: “Carona (sic) virus vaccine ready. Able to cure patient within 3 hours after injection. Hats off to US Scientists. Right now Trump announced that Roche Medical Company will launch the vaccine next Sunday, and millions of doses are ready from it.”
The post has accumulated over 161,000 views, been shared over 1,100 times and received more than 60 comments.
The Analysis
With the confirmed cases of COVID-19 nearing 3000 in Australia as of March 27, the search for a vaccine that could halt the prolonged spread of the potentially deadly virus is becoming more desperate.
A March 23 post purports to show the vaccine that could do just that, but the items pictured aren’t what the post claims them to be.
AAP FactCheck has identified at least three other posts making the same claims (here, here and here).
AAP FactCheck has traced the packaging labels “Sugentec” and “COVID-19 IgM-IgG” pictured in the post to a pharmaceutical company’s product website. The website, from Korean medical diagnostic tools creator Sugentec, explains that COVID-19 IgM-IgG product is a test for the human antibodies created when a person is infected with COVID-19.
“Antibodies can be found in the blood of those who are tested after infection [with COVID-19]. The antibodies detected by this test indicate that a person had an immune response to COVID-19, whether symptoms developed from infection or the infection was asymptomatic,” reads the website.
The product pictured on the website bears the same markings and labels as the one in the March 23 post.
AAP FactCheck has traced the image used in the March 23 post back to a highly shared Twitter post made by German journalist Florian Witulski in relation to a new test for COVID-19. The post made no mention of a vaccine. The post reads “Korea finished developing the 10 minute Covid-19 diagnostic kit and is now ramping up production. They plan to export 300.000 test-kits per week”.
The earliest instance of the image is in an article on a Korean website called Bio-Spectator,published March 20.
To date, there is no vaccine and no specific antiviral medicine to prevent or treat COVID-19, according to Australia’s Department of Health, the World Health Organization and the Centre for Disease Control. Around 35 organisations and countries, including Australia’s CSIRO are racing to create a vaccine for COVID-19, with one company entering human trials as of March 23, The Guardian and Time Magazine report.
The Verdict
Based on the evidence, AAP FactCheck found the Facebook posts to be false. Health authorities have stated there is currently no readily available vaccine for COVID-19. The photo in the March 23 post is in fact a testing kit used to detect COVID-19 antibodies..
False – The primary claims of the content are false.
* AAP FactCheck is accredited by the Poynter Institute’s International Fact-Checking Network, which promotes best practice through a stringent and transparent Code of Principles. https://factcheck.aap.com.au/
All information, text and images included on the AAP Websites is for personal use only and may not be re-written, copied, re-sold or re-distributed, framed, linked, shared onto social media or otherwise used whether for compensation of any kind or not, unless you have the prior written permission of AAP. For more information, please refer to our standard terms and conditions.

















