New products help doctors diagnose and treat patients within the cataract and corneal refractive workflows.
JENA, Germany and DUBLIN, Calif., May 3, 2023 /PRNewswire/ — ZEISS Medical Technology will showcase new surgical workflow innovations at the American Society of Cataract and Refractive Surgery (ASCRS) annual conference from May 6-8, 2023, in San Diego, CA. These products as part of the ZEISS Medical Ecosystem help enhance ophthalmic care within the cataract and corneal refractive workflows for improved diagnosis and treatment efficiency of surgical patients:
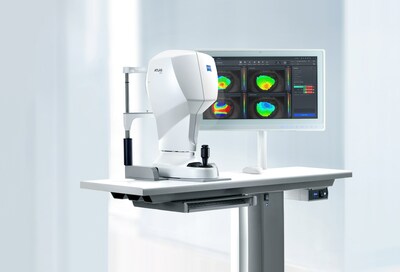
- The ATLAS™ 500 from ZEISS supports more efficient surgical diagnostics enhancing patient outcomes.
- The CT LUCIA® 621P Monofocal IOL from ZEISS received U.S. FDA approval, offering a unique ZEISS Optic to treat a wide range of cataract patients.
- With more than 7 million eyes treated with SMILE® worldwide, the ZEISS LVC portfolio helps create a high vision standard in laser vision correction.
- The VISULAS® yag laser from ZEISS delivers high-precision treatment for a high variety of IOL conditions.
“We are excited to offer these innovations to help maximize surgical workflow efficiency and performance, and further transform patient care in ophthalmology,” said Euan S. Thomson, PhD, President of the Ophthalmology Strategic Business Unit and Head of the Digital Business Unit for ZEISS Medical Technology. “ZEISS believes in a fully connected and integrated approach to eye care, and we continue to focus on the development of workflow solutions that will improve the quality of life for patients.”
Supporting more efficient surgical diagnostics for better patient outcomes
With more than 25 years of experience in corneal topography, the ATLAS™ 500 from ZEISS is the next-generation corneal topographer capable of the imaging and measurement of the cornea for assessment of various pathologies. The ATLAS 500 provides an intuitive and well-organized interface for efficient operation and workflow-optimized visualization. It integrates into both the corneal refractive and cataract workflows.
Offering a unique ZEISS Optic to treat a wide range of cataract patients in the U.S.
ZEISS delivers intuitive equipment and consumables to support reliable patient outcomes for a wide range of cataract patients. The CT LUCIA® 621P Monofocal IOL from ZEISS recently received U.S. FDA approval, featuring a patented ZEISS Optic (ZO) Asphericity Concept designed to compensate for a wide range of spherical aberrations and at the same time optimize visual outcomes in the event of potential decentration and lens misalignments. The ZEISS CT LUCIA 621P also comes in a fully preloaded injector for an intuitive, smooth, and controlled injection. Additionally, the architecture of the IOL is designed to enable easy centering while maximizing direct capsular contact, thus ensuring stability and supporting a consistent position in the bag.
“The line has been blurred between refractive and cataract surgery – for us surgeons that means we must always be raising our game to get patients the visual outcomes they expect. When I tell patients they are going to receive a ZEISS intraocular lens, ZEISS is a name they know and see as synonymous with quality giving them confidence in the product and the procedure. The ZEISS legacy in optics is great for surgeons because we know they make incredible lenses and great equipment too,” said Sydney L. Tyson, M.D., Ophthalmologist, Cataract & LASIK Surgeon, and Founder of Eye Associates & SurgiCenter of Vineland (USA).
Creating a high standard in laser vision correction
ZEISS celebrates a new milestone with more than 7 million eyes treated with SMILE® worldwide, building on its claim as a leader in laser vision correction with one of the most comprehensive LVC portfolios in the industry. The ZEISS portfolio creates a high vision standard while enabling surgeons to grow their business and provide broader choices and a viable solution in refractive surgery to any eligible patient. SMILE® is a minimally invasive corneal refractive procedure performed by using the VisuMax® femtosecond laser creating a thin disc-shaped lenticule within the cornea, which is then removed through a small incision, thereby achieving the desired vision correction.
Delivering high-precision treatment for a high variety of IOL conditions
The VISULAS® yag laser from ZEISS is a modern and compact laser workstation that offers high-precision treatment for safe and effective photodisruption. The laser uses an infrared light to perform posterior capsulotomy for a high variety of IOLs and peripheral iridotomy. Its high-precision super gaussian beam focuses laser energy offered in 22 levels with optimal gradation onto the point of treatment. Based on the selected application, the automated focus shift sets the focal point of the aiming beam precisely in front of, behind or directly at the focal point of the disruption laser.
ZEISS will showcase its new surgical workflow solutions during the American Society of Cataract and Refractive Surgery (ASCRS) Annual Conference from May 6-8, 2023, at booth 1437.
For more information, visit www.zeiss.com/med.
Not all products, services or offers are approved or offered in every market and approved labeling and instructions may vary from one country to another. For country-specific product information, see the appropriate country website. Product specifications are subject to change in design and scope of delivery as a result of ongoing technical development.
Contact for investors
Sebastian Frericks
Director Investor Relations
Carl Zeiss Meditec AG
Phone: +49 3641 220 116
Mail: investors.meditec@zeiss.com
Brief Profile
Carl Zeiss Meditec AG (ISIN: DE0005313704) is one of the world’s leading medical technology companies and is included in the German MDAX and TecDAX stock indices. The company supplies innovative technologies and application-oriented solutions designed to help doctors improve the quality of life of their patients. The company offers complete solutions for the diagnosis and treatment of eye diseases – including implants and consumables. In the field of microsurgery, the company provides innovative visualization solutions. With 4,224 employees worldwide, the company generated revenue totaling €1,902.8 million in fiscal year 2021/22 (ended September 30, 2022).
The company is headquartered in Jena, Germany. It has subsidiaries in Germany and abroad; more than 50 percent of its employees are based in the United States, Japan, Spain and France. The Center for Application and Research (CARIn) in Bangalore, India, and the Carl Zeiss Innovations Center for Research and Development in Shanghai, China, enhance the company’s profile in these rapidly developing economies. Around 41 percent of Carl Zeiss Meditec AG shares are in free float. The remaining approximately 59 percent are held by Carl Zeiss AG, one of the world’s leading companies in the optical and optoelectronic industries.
For further information visit: www.zeiss.com/med

SOURCE Carl Zeiss Meditec AG



















